Year :
2013
Title :
Chemistry
Exam :
JAMB Exam
Paper 1 | Objectives
31 - 40 of 44 Questions
| # | Question | Ans |
|---|---|---|
| 31. |
A primary amide is generally represented by the formula A. RCOOR B. RCONH 2 C. RCONHR D. RCONR 2 |
|
| 32. |
Which of the following fraction is used as raw material for the cracking process? A. kerosine B. lubricating oil C. bitumen D. diesel oils |
|
| 33. |
An organic compound contains 60% carbon, 13.3% hydrogen and 26.7% oxygen. Calculate the empirical formula A. C 5H 12O B. C 3H 8O C. C 6H 13O 2 D. C 4H 9O |
|
| 34. |
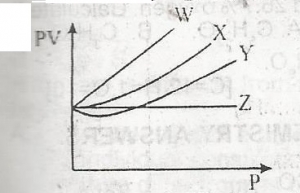 From the diagram above, an ideal gas is represented by A. Z B. W C. X D. Y |
|
| 35. |
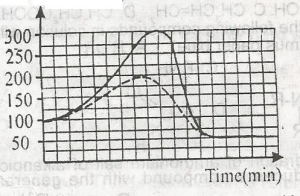 In the graph above, the activation energy of the catalyzed reaction is A. 100KJ B. 300KJ C. 250KJ D. 200KJ |
|
| 36. |
 Choose the correct option from the graph above . 3Fe(S) + 4H2O(g) ⇌ Fe3O4(s) + 4H2(g). The equilibrium constant, K, of the reaction above is represented as A. A B. B C. C D. D |
|
| 37. |
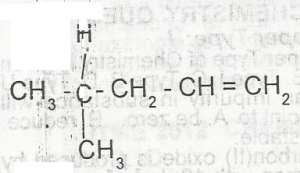 The IUPAC nomenclature for the compound above is A. 4-methylpent-1-ene B. 3-methylpent-2-ene C. 2-methylpent-1ene D. 2-methylpent-4-ene |
|
| 38. |
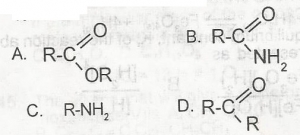 The dehydration of ammonium salt of alkanoic acids produces a compound with the general formula A. A B. B C. C D. D |
|
| 39. |
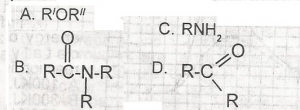 Which of the following compounds in solution will turn red litmus paper blue? A. A B. B C. C D. D |
|
| 40. |
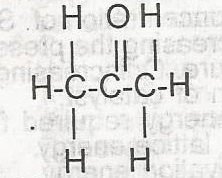 The compound above is an A. alkanone B. alkanoate C. alkanal D. akanol |
| 31. |
A primary amide is generally represented by the formula A. RCOOR B. RCONH 2 C. RCONHR D. RCONR 2 |
|
| 32. |
Which of the following fraction is used as raw material for the cracking process? A. kerosine B. lubricating oil C. bitumen D. diesel oils |
|
| 33. |
An organic compound contains 60% carbon, 13.3% hydrogen and 26.7% oxygen. Calculate the empirical formula A. C 5H 12O B. C 3H 8O C. C 6H 13O 2 D. C 4H 9O |
|
| 34. |
 From the diagram above, an ideal gas is represented by A. Z B. W C. X D. Y |
|
| 35. |
 In the graph above, the activation energy of the catalyzed reaction is A. 100KJ B. 300KJ C. 250KJ D. 200KJ |
| 36. |
 Choose the correct option from the graph above . 3Fe(S) + 4H2O(g) ⇌ Fe3O4(s) + 4H2(g). The equilibrium constant, K, of the reaction above is represented as A. A B. B C. C D. D |
|
| 37. |
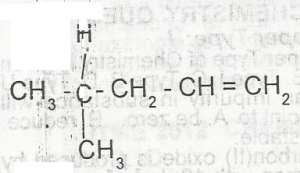 The IUPAC nomenclature for the compound above is A. 4-methylpent-1-ene B. 3-methylpent-2-ene C. 2-methylpent-1ene D. 2-methylpent-4-ene |
|
| 38. |
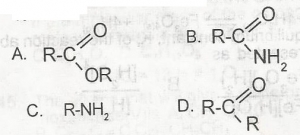 The dehydration of ammonium salt of alkanoic acids produces a compound with the general formula A. A B. B C. C D. D |
|
| 39. |
 Which of the following compounds in solution will turn red litmus paper blue? A. A B. B C. C D. D |
|
| 40. |
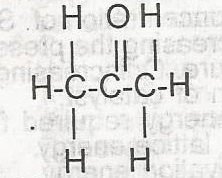 The compound above is an A. alkanone B. alkanoate C. alkanal D. akanol |